
Desvenlafaxine (which is Pristiq generic) is an antidepressant that is FDA-approved for the treatment of major depression in adults and has also been used off-label to treat vasomotor instability or “hot flashes” in menopausal women. Desvenlafaxine produces its therapeutic effects by inhibiting the neuronal reuptake of both Serotonin and Norepinephrine and therefore increases the levels of these neurotransmitters in blood. Although this effect is beneficial in alleviating the symptoms and signs of major depression, it also produces a broad range of Pristiq side effects.
Pristiq side effects include nausea, dizziness, insomnia, excessive sweating, constipation, and decreased appetite. Many patients experience these symptoms during the initial treatment phase, with effects diminishing over time. Sexual dysfunction also affects numerous users, contributing to treatment discontinuation.
Drug interactions of Pristiq includes dangerous combinations with alcohol, which intensifies neurological effects like dizziness and confusion. Concurrent use with medications such as Wellbutrin increases seizure risks, while combinations with Vyvanse or Adderall lead to sympathomimetic symptoms.
The dangers of Pristiq overdose are headache, vertigo, pupillary dilation, agitation, nausea, vomiting, and in severe cases, liver necrosis and serotonin syndrome. Patients experiencing persistent adverse reactions benefit from medical consultation rather than self-discontinuation.
Table Of Contents:
The emergence of these adverse reactions occurs because Serotonin and Norepinephrine beyond the regulation of mood are involved in the regulation of many other physiologic processes such as blood pressure, appetite, sexual function, and sleep among others. Therefore, increased serotonergic and noradrenergic activity will affect all these processes and will produce the associated side effects of Pristiq use.
This article will discuss the broad spectrum of Pristiq side effects, and additionally provide information about several possible drug-to-drug interactions and on the topic of Pristiq overdose.
Although the majority of patients will experience mild Pristiq side effects that are expected to abate with continued use, some patients may not tolerate Desvenlafaxine use well and may experience moderate to severe adverse reactions. This can greatly hinder patient adherence to medical therapy and delay the recovery of these patients.
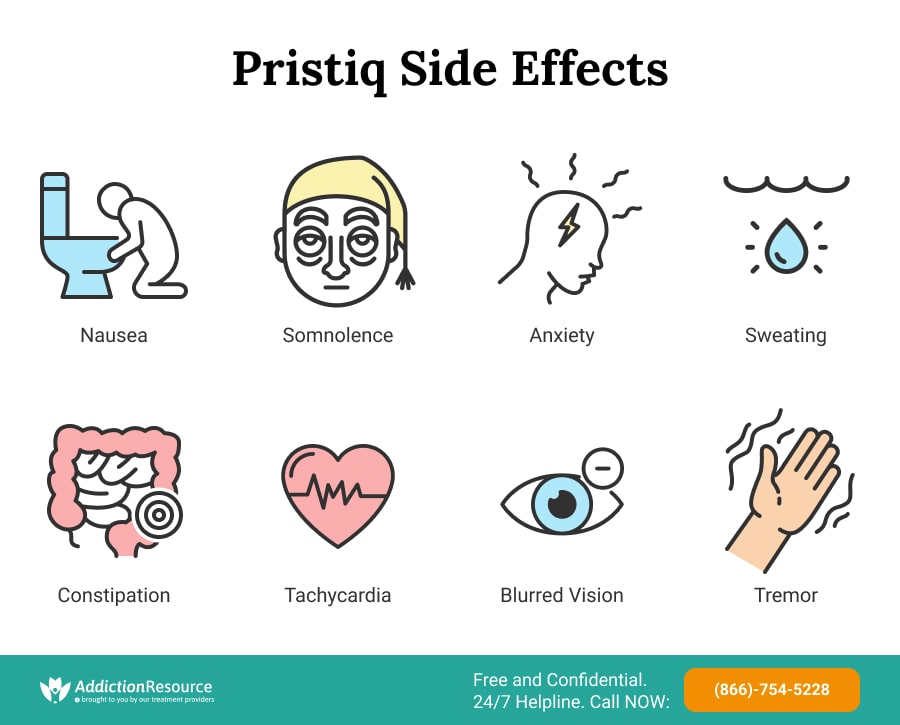
Common Pristiq Side Effects
As mentioned above, Pristiq is a medication used in Major Depressive Disorder (MDD) that works by inhibiting the reuptake of Serotonin and Norepinephrine, or as Serotonin Norepinephrine Reuptake Inhibitor (SNRI). Due to this mechanism of action, it increases the levels of these two neurotransmitters, and it is to this increase that the majority of Pristiq side effects can be ascribed to. Unfortunately, Desvenlafaxine has a broad list of adverse reactions that patients may experience, but fortunately, some of these reactions rarely manifest themselves, while others are experienced by the majority of patients and oftentimes abate with continued use.
Below Are Offered the Most Common Side Effects of Pristiq that Have an incidence of ≥5% as Reported by the FDA:
- Nausea
- Dizziness
- Insomnia
- Hyperhidrosis (increased sweating)
- Constipation
- Somnolence
- Decreased appetite
- Anxiety
- Specific male sexual function disorders
Outside of the most common side effects of Pristiq listed above, other frequently encountered adverse reactions affect multiple body systems. These adverse reactions include:
Constitutional Adverse Reactions
Constitutional effects associated with Pristiq use include those that affect an individual’s general well-being. These adverse constitutional effects include fatigue, chills, feeling jittery, and abnormal physical weakness or lack of energy.
Cardiovascular Adverse Reactions
One of the most concerning adverse reactions associated with Desvenlafaxine use is hypertension, which refers to increased blood pressure. This increase in blood pressure may be transient or sustained and can increase the user’s risk of hypertension-associated health complications. Other cardiovascular adverse effects include palpitations and tachycardia. Therefore it is important to disclose information about a prior history of elevated blood pressure to the prescribing medical professional before taking Pristiq and to alert them of treatment-associated elevation in blood pressure so that adequate dose adjustments can be made.
Gastrointestinal Adverse Reactions
Gastrointestinal symptoms outside of nausea and constipation can include dry mouth, vomiting, and diarrhea. These reactions are usually self-limited and only last for several days. However, If diarrhea and vomiting are experienced for a prolonged period of time, they can lead to variable degrees of dehydration, which can be life-threatening if not adequately addressed.
Treatment-associated gastrointestinal problems can be quite distressing to patients and knowing how to deal with these reactions is important. Nausea and vomiting can be dealt with by taking medications with a small amount of food. Some benefits can also be achieved by consuming ginger-containing foods and beverages. Persistent diarrhea or constipation requires medical attention and pharmacologic treatment with either antidiarrheal drugs or with laxatives and stool softeners.
Neurological And Psychiatric Adverse Reactions
Neurological and Psychiatric adverse reactions outside of dizziness, somnolence, insomnia, and anxiety listed above can include headache, blurred vision, mydriasis, vertigo, tinnitus, tremor, paraesthesias, disturbances in attention, irritability, nervousness, and abnormal dreams.
Adverse effects such as dizziness, somnolence, and insomnia can be particularly distressing for patients and can be major contributors to drug discontinuation. Therefore, help must be sought for these problems. Management strategies can range from decreasing caffeine intake, evaluation of sleep patterns and counseling on sleep hygiene measures, adopting exercise regimens, altering the dose or changing the dosing schedule of Desvenlafaxine, switching to a different antidepressant, to using adjunct drugs.
Uncommon And Rare Pristiq Side Effects
Uncommon reactions refer to those that occur in approximately 5 people out of 1000 people given a particular medication, and rare reactions are those that occur in approximately 5 people out of 10,000 people given a particular medication. Classifying adverse reactions as uncommon or rare is quite challenging as the frequency of their occurrence is difficult to trace without extensive medical studies that provide information about the exact prevalence of such reactions.
Possible Uncommon and Rare Side Effects of Pristiq Can Include:
- Abnormal bleeding
- Activation of episodes of mania or hypomania
- Renal impairment
- Difficulty Urinating
- Convulsions and Seizures
- Hyponatremia
- Interstitial lung disease and eosinophilic pneumonia
- Extrapyramidal disorder
- Depersonalization
- Bruxism
- Dysgeusia or loss of taste
It is important to remember that this is not an exhaustive list of all possible uncommon reactions and if any of them are experienced it is important to seek immediate medical care. Also, symptoms of withdrawal from Pristiq are quite rare when the dose is reduced correctly. Rapidly cutting down or stopping the medication may result in irritability, nightmares, vomiting, confusion, fatigue, and suicidal thoughts.
The FDA does not report significant changes in appetite and body mass in patients who use this drug, thus the topic of Pristiq and weight gain is ambiguous.
Pristiq And Weight Gain
The phenomenon of Pristiq and weight gain is quite a bothersome effect of treatment and it is one of the most important factors that interfere with treatment adherence. Weight gain associated with antidepressant use is quite a complex topic as its origin is unclear. It can be a sign of improvement in a patient who had experienced weight loss as a symptom of Major Depressive Disorder. It can be a leftover phenomenon from atypical depression, or it can be something independently related to depression or Desvenlafaxine itself.
Regardless, if users do experience weight gain that is unacceptable to them several management strategies exist. Primarily dietary and physical activity interventions should be attempted. If these methods fail the next steps should be done under the guidance of a medical doctor. First, a dose adjustment should be attempted, and if this is also ineffective switching to different drugs with lower propensities for weight gain can be considered.
Pristiq Sexual Side Effects
Pristiq sexual side effects are incredibly common. Antidepressants, including Desvenlafaxine, typically affect libido, arousal, orgasm, and ejaculation and may also affect vaginal lubrication and erection. These adverse effects also seem to occur more frequently at higher doses of the drug, and as such can be said to be dose-dependent.
The Most Commonly Occurring Sexual Side Effects of Desvenlafaxine May Include:
- Anorgasmia
- Decreased Libido
- Abnormal Orgasm
- Erectile Dysfunction
- Delayed Ejaculation
- Ejaculation Failure
- Sexual Failure
Sexual side effects of Pristiq can be immensely frustrating for all patients regardless of gender. Some people discontinue the treatment due to this influence of the drug, tumbling into depression again and losing their achieved progress. Therefore a doctor must be consulted so that appropriate steps can be taken to manage associated dysfunction. These steps can include dose reduction, the timing of sexual activity toward the end of a dosing interval, drug holidays, addition of “antidote” drugs, and switching to agents with lesser degrees of sexual dysfunction.

Pristiq Side Effects During Pregnancy And Breastfeeding
Pristiq has pregnancy category C, which means that there is not enough information to ensure the drug’s safety concerning the health of pregnant women and their babies. Although it is generally not recommended to take antidepressants such as Pristiq during pregnancy due to the risk of potential adverse effects, sometimes the drug is necessary. Only a doctor can decide whether the benefits of using the medication outweigh the potential risk to pregnancy and/or breastfeeding.
Taking desvenlafaxine while breastfeeding is generally safe for the infant’s health, although the child needs to be monitored and examined frequently. Studies indicate that infants ingest negligible doses of the drug with their mothers’ milk. The concentration of the medication in a child’s plasma is approximately 4.8% of the concentration in the mother’s plasma. It is necessary to contact a doctor if the child becomes excessively sleepy or develops weight gain anomalies.
Pristiq Interactions: The Possible Trigger For Adverse Effects
As with all medications, Desvenlafaxine is subject to drug-to-drug interactions (DDIs) as well. These interactions refer to changes in the action or side effects of Pristiq caused by its concomitant administration with food, beverage, supplement, or other drugs. Thus DDIs can predispose users to tolerability issues, problems with lack of efficacy as well as serious adverse events. Below are discussed select few DDIs that Pristiq can be subject to.
Pristiq And Alcohol
Ethanol, the organic chemical contained in Alcohol, and Desvenlafaxine are both drugs that mediate their effects in the central nervous system. Therefore, concomitant use of Pristiq and alcohol can lead to an experience of increased neurologic and cognitive effects of this medication such as:
- dizziness
- somnolence
- confusion
- impairment of attention and psychomotor skills
Thus, it is best to completely avoid or limit the consumption of alcohol while taking Desvenlafaxine. Additionally, in the case that users do combine the use of Pristiq and alcohol they should avoid all activities that require mental alertness and motor coordination such as driving or operating machinery. For more information about the DDIs of Pristiq and alcohol, users are encouraged to contact their prescribing physicians.
Pristiq And Wellbutrin
Bupropion (Wellbutrin) is an atypical antidepressant used to treat Major Depression and to help smoking cessation. Bupropion is unfortunately associated with a dose-related risk of seizures. Combining this medication with Desvenlafaxine, which also has a small risk of causing seizures, can greatly increase the overall risk of a user experiencing a seizure. Thus the combined use of these two epileptogenic medications poses a major health risk to users and should be avoided. However, if the coadministration of these two medications is necessary, it should only be done under the supervision and guidance of the prescribing physician.
Pristiq And Vyvanse
Desvenlafaxine and Vyvanse (Lisdexamfetamine) are another combination that should be avoided due to the possible DDIs. Lisdexamfetamine is an amphetamine drug that is used to treat signs and symptoms of Attention-Deficit Hyperactivity Disorder (ADHD) and Binge Eating Disorder (BED). Unfortunately, SNRIs, like Pristiq, can exaggerate some of the sympathomimetic effects of Vyvanse and produce symptoms such as jitteriness, racing thoughts, palpitations, tremors, dry eyes, stomach cramps, and restlessness. Furthermore, as both of these agents possess serotonergic activity, their co-administration could result in Serotonin syndrome, a rare but serious and potentially fatal syndrome associated with autonomic instability. Therefore, the use of these two medications together is discouraged.
Desvenlafaxine is generally safe and effective, and, in some cases, it can be used during pregnancy under a doctor’s supervision.
Pristiq And Adderall
A combination of Pristiq and Adderall, which is a combination of amphetamine and dextroamphetamine used in the treatment of Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy, is also contraindicated for the same reasons as mentioned above. Desvenlafaxine can potentiate the sympathomimetic symptoms of Adderall and their combined serotonergic activities can increase the risk of experiencing Serotonin Syndrome in users.
Pristiq And Lamictal
Lamictal is the brand name for Lamotrigine which is a type of anticonvulsant medication that has a broad spectrum of use in seizures. The concurrent use of these two medications is contraindicated for two reasons. One, both of them are capable of causing low sodium levels in blood or hyponatremia, which can present as nausea, vomiting, headache, lethargy, muscle weakness, and spasms. Thus their concurrent use can increase the risk. Two, as Devenlafaxine is mildly epileptogenic it can reduce the efficacy of Lamictal and as such should not be used concomitantly.

Pristiq Overdose
The FDA reports limited clinical experience with Pristiq overdose as a standalone agent. However, the risk of overdose and acute intoxication increases when this medication is combined with alcohol and/or other drugs. Symptoms and signs of overdose can include:
- Headache
- Dizziness and Vertigo
- Pupillary dilation
- Agitation
- Change in the level of alertness
- Nausea and Vomiting
- Constipation
- Diarrhea
- Dry mouth
- Paresthesia
- Electrocardiographic changes
- Serotonin syndrome
- Liver necrosis
It is important to remember that overdose is uncommon if this medication is used as directed. However, in the case of overdose users should seek immediate emergency medical help. Unfortunately, there is no specific treatment for Desvenlafaxine overdose, but symptomatic and supportive treatment will be provided.
Do The Side Effects Of Desvenlafaxine Go Away?
Pristiq side effects go away without treatment in the majority of cases. Common and mild conditions, such as headache, nausea, and constipation, often resolve without medical intervention. If any of the effects persist and cause a person significant distress, it is important to contact their healthcare professional and determine whether it’s necessary to modify the treatment. Diarrhea and vomiting are particularly dangerous over a prolonged period of time.
Besides, some patients may be resistant to treatment or find the effects of the medication too insignificant. In this case, the person’s doctor might recommend switching from Pristiq to Effexor. The latter has a stronger acting substance and slightly different adverse effects, which may be beneficial to certain patients.
Never Neglect Side Effects Of A Medication
Desvenlafaxine is generally safe and effective, and, in some cases, it can be used during pregnancy under a doctor’s supervision. The most common adverse effects of Pristiq include nausea, constipation, increased anxiety, trouble sleeping, excessive sweating, and sexual problems. Normally, mild adverse symptoms go away on their own as the person gets used to the medication.
Some symptoms, however, are serious and require immediate medical attention. If any of these effects occur, the person should seek help as soon as possible and discuss other treatment options with their healthcare professional. Because of the sudden stop taking medication to self-medicate the adverse effects, they can become even worse because of withdrawal. That is why it is critical to consult with a doctor or in an addiction treatment center.
Hope Without Commitment
Find the best treatment options. Call our free and confidential helpline
Most private insurances accepted
Find Drug Rehabilitation Centers Near You Anywhere In the US
Addiction Resource team has compiled an extensive list of the top drug rehabilitation facilities around the country. Use our locator tool to find the best centers near you.
Page Sources
- Alibegović, A., Kariž, S., & Volavšek, M. (2019). Fatal overdose with a combination of SNRIs venlafaxine and duloxetine. Forensic science, medicine, and pathology, 15(2), 258–261. https://doi.org/10.1007/s12024-019-00097-3
- Atkinson, S., Lubaczewski, S., Ramaker, S., England, R. D., Wajsbrot, D. B., Abbas, R., & Findling, R. L. (2018). Desvenlafaxine Versus Placebo in the Treatment of Children and Adolescents with Major Depressive Disorder. Journal of child and adolescent psychopharmacology, 28(1), 55–65. https://doi.org/10.1089/cap.2017.0099
- Boyer, Patricea; Montgomery, Stuartb; Lepola, Ullac; Germain, Jean-Micheld; Brisard, Claudined; Ganguly, Ritae; Padmanabhan, Sudharshan K.e; Tourian, Karen A.e Efficacy, safety, and tolerability of fixed-dose desvenlafaxine 50 and 100 mg/day for major depressive disorder in a placebo-controlled trial, International Clinical Psychopharmacology: September 2008 - Volume 23 - Issue 5 - p 243-253 doi: 10.1097/YIC.0b013e32830cebed
- Darlene C. Deecher, Chad E. Beyer, Grace Johnston, Jenifer Bray, S. Shah, M. Abou-Gharbia and Terrance H. Andree (2006) Desvenlafaxine Succinate: A New Serotonin and Norepinephrine Reuptake Inhibitor: Journal of Pharmacology and Experimental
- Therapeutics 318 (2) 657-665; DOI:https://doi.org/10.1124/jpet.106.103382
- Kelly, K., Posternak, M., & Alpert, J. E. (2008). Toward achieving optimal response: understanding and managing antidepressant side effects. Dialogues in clinical neuroscience, 10(4), 409–418. https://doi.org/10.31887/DCNS.2008.10.4/kkelly
- Low Y, Setia S, Lima G. Drug–drug interactions involving antidepressants: focus on desvenlafaxine. Neuropsychiatr Dis Treat. 2018;14:567-580 https://doi.org/10.2147/NDT.S157708"
- Naseeruddin R, Rosani A, Marwaha R. Desvenlafaxine. [Updated 2021 Jul 15]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK534829/
- Sansone, R. A., & Sansone, L. A. (2014). Serotonin norepinephrine reuptake inhibitors: a pharmacological comparison. Innovations in clinical neuroscience, 11(3-4), 37–42.
- Santarsieri, D., & Schwartz, T. L. (2015). Antidepressant efficacy and side-effect burden: a quick guide for clinicians. Drugs in context, 4, 212290. https://doi.org/10.7573/dic.212290
- Wyeth Pharmaceutical Inc. (2011, July). HIGHLIGHTS OF PRESCRIBING INFORMATION FOR PRISTIQ® (desvenlafaxine). Www.Accessdata.Fda.Gov. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/021992s030lbl.pdf
- Zhong, Z., Wang, L., Wen, X., Liu, Y., Fan, Y., & Liu, Z. (2017). A meta-analysis of effects of selective serotonin reuptake inhibitors on blood pressure in depression treatment: outcomes from placebo and serotonin and noradrenaline reuptake inhibitor controlled trials. Neuropsychiatric disease and treatment, 13, 2781–2796. https://doi.org/10.2147/NDT.S141832


 Reviewed by:
Reviewed by:  Written by:
Written by: 
 FindTreatment.gov
FindTreatment.gov