
Opana withdrawal can be quite severe, causing a long list of symptoms that become conspicuous when the body tries to carry out its regular functions without the drug. The highly potent, last-resort pain medication changes the homeostasis in the brain. Due to the habit-forming characteristics of the drug and the possibility of one experiencing withdrawal symptoms to the medication, this drug is only administered when other pain relievers have failed to provide relief. Extreme cases of Opana withdrawal are also commonly experienced by individuals who abuse the opioid narcotic for recreational purposes.
Table Of Contents:
- What is Oxymorphone?
- What is the History of The Drug?
- What Are Some Side Effects of Opana?
- Which Opioid is More Potent: Opana or Oxycodone?
- How Addictive is Oxymorphone?
- What are the Signs of Abuse?
- How Long Does Opana Stay in Your System?
- What are Opana Withdrawal Symptoms?
- What are Opana Addiction Treatment Techniques?

Let’s take a look at the mechanism of Opana withdrawal, its pharmacokinetics, health issues associated with the misuse of the opioid and addiction treatment techniques.
What is Opana Drug?
Oxymorphone is a controlled opioid explicitly used for relief of intermediate to severe pains. The opioid treatment is typically recommended when other similar pain medications are ineffective for the condition. Oxymorphone ER belongs to the category of narcotic analgesics, as it is known to act directly on the central nervous system (CNS) to relieve pain. Most of the side effects experienced after use is due to their effect on the CNS.
Opana is the brand name for the Oxymorphone formula, similar in its effects to Morphine. It comes in both immediate- and extended-release tablets.
How Opana Works in the Body
Opana medication exerts pharmacologic effects on both the central nervous system and the gastrointestinal tract. Oxymorphone binds to the MORs (mu-opioid receptors) in the brain that yields intense analgesic effects. The principal actions of Opana medication are sedation and analgesia. Opana helps the brain lessen pain anywhere in the body. Excessive dosage can lead to a feeling known as an anxiolytic impact. Opana drug also binds to and hinders GABA inhibitory interneurons through mu-receptors. These actions modulate pain signals.
Consuming Oxymorphone with food can lead to an increase in blood serum levels up to 50%. Furthermore, consuming it with alcohol can increase blood serum levels by up to 270%. For this reason, it’s advisable to ingest the tablets an hour to an hour and a half before meals and to avoid alcohol consumption during the treatment course.
More About Opioids:
Opana Drug Class
Oxymorphone, the semisynthetic opioid, is related to hydromorphone, a derivative of Morphine. The DEA classifies Oxymorphone as a Schedule II controlled substance. Categories of drugs under schedule II Substance are those that affect the central nervous system. The narcotic analgesic is used to suppress moderate to extreme pain such as post-surgery and others.

Opana History
Oxymorphone was initially manufactured for medical use in 1959 as injectable and rectal suppository forms.
The oral drug appeared on the market in the US in 2006 manufactured by Endo Pharmaceuticals. It was presented in IR and ER forms and was twice as potent as OxyContin.
Opana’s addictive nature has led to an increase in pharmacy thefts since the reformulation of OxyContin in 2010. Narconon reports that 33 deaths occurred in Louisville, Kentucky, in 2011. This was due to an Opana overdose.
In 2012, an estimated 1 million Opana prescriptions was written. The powerful adverse effects and withdrawal risks of the medication prompted the DEA to classify the medication as a schedule II substance.
Endo Pharmaceuticals reformulated Opana in 2012 after the FDA request. The reformation was geared towards stopping abuse by making the Opana pill extremely hard to crush; this way, it would be difficult to crush as inhaled through the nasal route. The FDA’s decision was based on the trending shift in the route of abuse of the medication.
Unfortunately, this led to injection abuse of the reformed Opana ER. The risks of the Opana pill included an outbreak of hepatitis C and HIV in 2015 due to the continued sharing of needles by abusers. That outbreak pushed FDA to make valid strides in protecting the public from exposure and potential for misuse of Opana medication.
June 08, 2017, marked the end of the distribution of Oxymorphone ER in the US market as the Food and Drug Administration instructed the removal of Opana drug from the market for health reasons. Due to its addictive nature, the FDA asked that Endo Pharmaceuticals no longer offer its Opana Extended Release; and Endo removed the medication from the market.
The FDA emphasized that ending the opioid epidemic required active steps to end the public health crisis.
However, regular Opana is still on the market, and so are generic extended-release tablets. Oxymorphone ER is currently available under a highly restricted and monitored distribution program referred to as Opioid Analgesic REMS (Risk Evaluation and Mitigation Strategy).
There have been lawsuits on the production of Opana ER, as bureaucratic settlements were allegedly made to high-tier pharmaceutical companies to stop producing generic and affordable Oxymorphone ER medications over a while, granting a monopoly to certain pharmaceutical companies. The pay-for-delay agreement increased the scarcity of the drug and therefore increased demand.
Opana Side Effects
Due to its high potency, Opana drug most often presents with side effects, especially in abusers of the medication. The following Opana side effects occur in 1% to 10% of the patients. Indeed, they include:
- Blurred vision
- Diarrhea
- Abdominal pain
- Indigestion
- Dry mouth
- Reduced hunger
- Physical weakness
- Fever
- Swelling
- Loss of weight
- Sleep problems
- Anxiety
- Mental confusion
- Restlessness
- Nervousness
- Depression
- Breathing difficulties
- Burning sensation and redness on the skin, especially of the face and neck
- Elevated blood pressure
Less common Oxymorphone side effects may include:
- Fullness in the stomach
- Symptoms of allergy
- Slowed pulse
- Altered consciousness
- Urinary problems
- Sense of well-being or feeling down
- Worry
- Hallucinations
- Low blood pressure
- Decreased mental functions
- Racing heartbeat
- Reduced pupil size
- Decreased breathing
- Loss of consciousness
- Difficulty passing urine
- Skin rashes
Symptoms Of Allergy To Oxymorphone
Allergic reactions are the results of an abnormal response to the immune function. They can be against Oxymorphone itself or any ingredient in the preparation. In any case, the immune cells mistake it for a foreign particle. As a result, they launch an abnormal immune attack. Opana allergy is rare. Nevertheless, it warrants emergency medical care.
Watch for the Following Symptoms of Allergy:
- Itching
- Rashes
- Severe dizziness
- Severe breathing problems
- Swelling of the face, tongue, or throat
Opana Overdose Symptoms
How can oxymorphone overdose be detected? The symptoms of overdose may include clammy skin, slow heart rate, muscle weakness, pinpoint pupil, slow and paused breathing, extreme drowsiness, confusion, and possible coma. A person who may have overdosed on Opana may look pale with blue colored lips, and may be unconscious.
In this case, the first line of action is to call an emergency hotline 911 or seek medical care immediately as further delay may lead to death.
Opana vs. Oxycodone: Which Is More Potent?
Well, it is not an easy question to answer. Both these medications are very strong. Also, they belong to the same class of pain medications. For these reasons, the patients and their caretakers often have this question in mind.
Whether Oxymorphone may seem stronger than Oxycodone because it requires a lower amount to produce comparable pain relief, the following characteristics should also be considered:
- The degree of pain. For acute pain, Oxycodone is a more common drug to take. Oxymorphone is prescribed when the pain is severe.
- Time to produce pain relief. Oxymorphone is more fat-soluble than Oxycodone. That said, it reaches the brain faster. As a result, one gets more rapid pain relief.
- The lower number of adverse reactions. Opana ER produces fewer side effects than Oxycodone CR.
- When medical professionals calculate the equianalgesic dose of the Opana pill, they find that the medication has a bit less to worry about, as opposed to its competition. Opana ER causes fewer problems with memory and focus. Also, a lower degree of sedation.
- The ratio of the doses required to produce equivalent pain relief. It is called an equianalgesic dose. Studies suggest the equianalgesic dose of Oxycodone CR on Opana ER is 2:1. It means Oxymorphone in half the Oxycodone dose produces similar effects. Oxymorphone is stronger than Oxycodone. It is even more potent than the parent medication Morphine. Depending on the severity of the pain and the counsel of a medical professional, it may be better and more cost-effective to opt for a lower dose of the Opana pill than Oxycodone CR.
Note that only an experienced doctor should calculate the equianalgesic dose. Similarly, one should not take this information as a substitution for the doctor’s advice. Take care not to think Oxymorphone is two times stronger than Oxycodone because of the ratio. Medical calculations are more than pure mathematics.
A patient should know that individual response to medication may vary. Particularly, this is more familiar with opiate therapy. Thus, they should not generalize any health information. In a nutshell, everyone is unique, and so is the pain. Only a doctor can decide which medication is better for the health needs.
Opana Addiction Dangers
Oxymorphone is easily abused because it changes the brain chemistry by increasing the “pleasure” feel and induces relaxation while numbing pain. The individual may also begin to experience flu-like withdrawal symptoms that can escalate into severe health complications.
Opana is very addictive and habit-forming in nature. Even if one takes prescribed amounts of opioid drugs such as this one regularly, it may cause the user to tolerate it. As a result, it leads the user to take a higher dose of Oxymorphone to achieve the same effect.
A patient with a personal history of substance abuse is more likely to become heavily dependent on Opana. The risk groups of Oxymorphone ER are those who are most vulnerable to its effects and would most likely experience health complications during use. These individuals include alcoholics, pregnant women, people with respiratory conditions, patients who take benzodiazepines, and children.
Accidental ingestion of even a single oxymorphone ER dose can be fatal for children. In pregnant women, it can lead to Neonatal Opioid Withdrawal Syndrome.
Except for the dangers mentioned above, the opioid crisis in the US shows the impressive numbers of people suffering from painkillers abuse.
A 2016 national survey by the Substance Abuse and Mental Health Services Administration (SAMHSA) on public health and drug use showed that 18.7 million people misused opioids in the oxymorphone category. Similarly, in 2016, about 2.1 million people within the age of 12 and older were diagnosed with an opioid use disorder.
At least three million citizens of the United States and 16 million worldwide are addicted to opioids. Dependence and abuse of the medication are particularly prominent within age groups 26 and older.
Reports from the National Survey on Drug and Health (NSDUH) showed that about 4.3 million people aged 12 and above were currently abusing Oxymorphone painkillers.
Another report from the National Institute on Drug Abuse (NIDA) stated that at least 2 million people in the US suffered from prescription opioid addiction.
In addition, the NSDUH reported that the demographic implicated as the highest users of prescription opioids are individuals within the age range 18 to 25.

Signs and Symptoms of Opana Abuse
Oxymorphone is an opioid, so the signs and symptoms of Opana abuse are similar to other opiates such as heroin. These can include:
- Nausea
- Vomiting
- Muscle aches
- Euphoria
- Changes in blood pressure
- Extreme relaxation
- Drowsiness
- Respiratory difficulties
- Constipation
- Constricted pupils
- Tremors
- Twitches
- convulsions
Behavioral signs that an individual might have a problem with Opana abuse are:
- The person looks for a reason to take it, even when it is not necessary
- Difficulty in focusing on anything but their drug
- Always having the drug on them anywhere they go
- Often hides the drug in secret places
- Spending all of their money and borrowing to buy medications
- Oxymorphone use takes priority, even on attending to responsibilities
- The person repeatedly claims that he or she has lost their prescription
- Exaggerating about the levels of pain he or she experiences when consulting a doctor trying to get a prescription for the drug
- Tampering with a prescription
- Obtaining a prescription for the drug from more than one doctor
- Purchasing this drug over the internet without a prescription
- Using it with alcohol or other drugs
- Developing a tolerance and requiring higher or more frequent doses
The only effective solution to deal with an Opana addiction is to get the person into a rehabilitation program as soon as possible. Rehabilitation from this drug will typically last between eight and ten weeks.
Unfortunately, drug addiction doesn’t only affect the user physically. However, it also leaves them with relationships to repair and self-respect to regain. They should learn new life skills, which will replace the harmful ones.
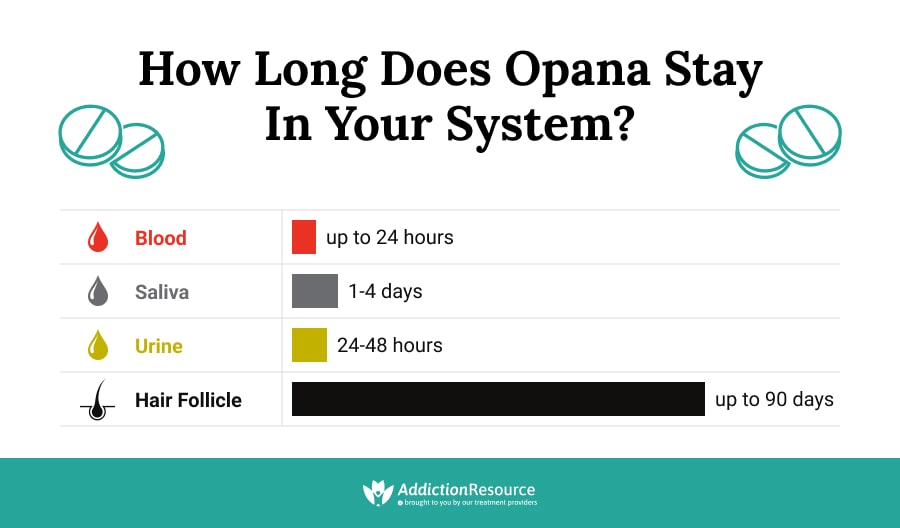
How Long Does Opana Stay in Your System?
The drug has a relatively long half-life of 8 hours, compared to Morphine which has a half-life as short as 1.5 hours. Giving exact duration can be hard because there are so many factors involved. One can administer the drug either in immediate-release (IR) or extended-release (ER) format. Immediate-release half-life is between 7.3 and 9.4 hours, and when taken in extended-release, the half-life can be between 9 and 11 hours.
In case of immediate release, the drug can take from 1.67 days to 2.15 days to exit the system. Also, in the case of extended-release, it can take from 2 to 2.5 days to exit the system entirely.
How Long Does Opana Stay in the Urine?
A urine test involves taking a fresh sample from the person and testing the metabolites of Oxymorphone. The two primary metabolites are 6-OH-Oxymorphone and Oxymorphone-3-Glucuronides (O3G). Since O3G can be found in abundance in the urine of an Opana user, it’s analyzed using certain scientific techniques. A common one used is High-Performance Liquid Chromatography (HPLC).
The detection window of Oxymorphone metabolites in urine is from 24 to 48 hours. However, in some extreme cases, the metabolites can be detected for up to five days after the last dose.
Urine is mostly used for laboratory tests. However, Oxymorphone can also be detected in blood, hair, and saliva. It may be detected in the hair for 90 days and in the blood for about 24 hours after the last use. A saliva test can detect Opana in about 1 to 4 days.
A urine test is pretty standard to detect the use of opioids, and Opana can result positively in it.
Factors that Determine the Time for Opana to Stay in The System
Numerous factors determine how long an Opana drug will stay in one’s system. The human body is highly complex, and with each individual, the processes of pharmacokinetics may differ. Here are some factors that influence how long Oxymorphone stays in the body.
Method of Administration
Oxymorphone route of exposure contributes to its delayed presence in the body. Oral administration is the most popular route of exposure but less effective than other routes such as intramuscular. An Oxymorphone epidural injection lasts longer in the body than IV injections and IM. Similarly, a rectal suppository as well as Intramuscular injection is usually administered in individuals living with postoperative pains. Each route takes different periods of time to break down.
Type of the Drug Administered
One major factor that decides how long Opana stays in the system is the type of drug taken. IR is faster as the drug peaks in the serum within half an hour after the ingestion. However, this also results in the drug leaving your system via urine faster. On the flip, ER provides a slow but steady release and lasts much longer. So Oxymorphone ER takes longer to exit the system. According to careful estimates, Opana leaves the body about 10 hours faster when delivered as IR rather than ER.
Dosage
After the administration (IR or ER) format, the second major factor is the dosage taken. Pills are usually administered in the range between 5 and 40 mg. However, a higher dosage increases the workload of the body’s system while it is trying to excrete Oxymorphone. Also, it impacts efficiency.
Frequency and Duration of Taking the Drug
More frequent users develop tolerance to Opana, which means their body will take longer to get rid of the drug. On the flip, infrequent users are less likely to have the drug stored in their fat cells or renal pathways. Moreover, infrequent use also reduces the likelihood of dependence or tolerance development.
Personal Differences
Every person is different, and the bodies respond to drugs differently. For instance, two people who took the same dosage will vary regarding how long the drug stayed in their bodies. Underlying factors include people’s age, body mass, renal function, health, underlying conditions, food intake. For instance, elderly or obese people usually have a slower metabolism.
Other Drug and Alcohol Use
If one took Oxymorphone with alcohol, it could increase the plasma concentration of the drug. Typically it will increase as much as up to 270%. Alcohol strength plays a key role, and a higher volume of alcohol will increase the concentration. Several other drugs react the same way. The result is that half-life of the metabolites increases, and they can stay in the body for much longer.
Dependence on any drug can be dangerous, even life-threatening. Never take drugs without a prescription. Likewise, as soon as one feels they are taking more than prescribed or more often than recommended, they should instantly consult a physician.
Opana Withdrawal Symptoms
Being addictive, Opana can be taken for longer and larger doses than prescribed. This can lead to long-term problems and damage the body. The sooner a person abusing Oxymorphone starts to get clean, the more comfortable the detox process will be.
Withdrawal is the reaction of the mind and body to not having a substance to which it has grown accustomed. Opana withdrawal can be hard with how addictive it is, both physically and psychologically. Opana withdrawal, like other opioids, requires detox and medical professionals’ assistance to monitor the person’s condition. Detoxing from opioids comes with many risks, including problems with cognitive functions, respiratory system shut down, cardiac problems, and ultimately coma and death. Drug withdrawal typically begins within 14 to 18 hours after taking the last dose.
According to the FDA Drug Report, Opana Withdrawal Symptoms Include:
- Chills
- Sweating
- Restlessness
- Yawning
- Lacrimation
- Nausea
- Vomiting
- Restlessness
- Insomnia
- Joint pain
- Muscle pain
- Backache
- Weakness
- Abdominal pain
- Rhinorrhea (a runny nose)
- Hypertension
- Irritability
- Irregular or heightened heart rate
- Anorexia
- Pupil dilation
- Increased respiration
How Long Does Opana Withdrawal Last?
Opana stays in the system for days and can affect the mind and body during that time. Opana withdrawal symptoms can happen the whole length of this period. The typical detox path is that the symptoms will begin with mild agitation and confusion and get worse, building up to more severe and dangerous physical symptoms.
Day 1 to 3
The agitation and confusion symptoms will begin first and last the first one to three days. There is no clear point where a person starts one stage of withdrawal symptoms as everyone’s neurochemistry is different, and they start slowly, and as one may begin to fade or become less important, the others become worse and take precedence.
Day 3 to 6
The more severe symptoms will usually happen between days three and six. They will generally begin to fade after day seven as Oxymorphone leaves the system entirely. That is how long Opana withdrawal lasts typically.
How To Ease Opana Withdrawal Symptoms?
Unfortunately, there is nothing one can do to shorten the length of Opana withdrawal symptoms. What can be done is to provide relief from the symptoms and alleviate some of the sufferings they experience. This, of course, starts with stopping the use of Oxymorphone as soon as possible.
Then there is using some minor medications to help alleviate some of the agitation and aches and pains. Over-the-counter medications and vitamins, such as vitamin B, are standard treatments. Increasing the number of fluids, especially those with electrolytes, will help flush the system and keep the person hydrated. Finally, there are prescription medications to use which will help alleviate some of the Opana withdrawal symptoms. These can include medications to help with anxiety, things to help with sleep, or other drugs that help alleviate some cravings and withdrawal, such as methadone treatment. These would likely be used sparingly, as they may also have abuse potential.
Medications that May Be Used for Limiting the Opiate Withdrawal Timeline Include:
- Opioid agonists
- Tapering medications
- Benzodiazepines
- Opioid antagonist
- Antidepressants
- Partial antagonists and agonists
- Non-opioid agonists

Opana Addiction Treatments
Opana addiction treatment isn’t an easy process. Breaking out of the bonds of addiction and achieving full recovery is hard work. However, it can be done, and the results are rewarding.
Rehabilitation treatment care focuses on both getting the user off the drug and helping them remain in recovery. This means addressing the underlying causes of their addiction and building essential skills to help the user avoid relapse. The sooner treatment is started, the better the results tend to be.
Many people worry rehabilitation programs do not work. Everyone knows of someone who has been in and out of rehabilitation more times than they can count—if not someone they know personally, then a celebrity. This makes us think rehabilitation for Oxymorphone, and other drugs isn’t useful, but the research says otherwise. In general, when users go in with a positive or hopeful mindset, recovery programs work.
And these programs are not one-size-fits-all. Many people think rehabilitation is not for them because they have a specific idea of what it will be, and what they are picturing won’t work for them. In truth, there are many different Opana addiction treatment options available for users to choose from, allowing them to select one which is truly a good fit for them.
There are different types of therapies used, different locations, long-term and short-term options, and residential and outpatient programs. Ultimately, the right program is out there.
What are the Opana Rehab Programs?
There are numerous Oxymorphone rehabilitation programs in the United States. Because opioid addiction is, unfortunately, widespread, the majority of clinics can treat Opana addiction. This means the hard part is not finding a center for treatment but finding the one best suited to the user. Recovery from Opana addiction is more likely to be successful if the user is happy with the program they are enrolled in.
Some Factors to Keep in Mind When Selecting a Program Are:
- Residential or Outpatient: In general, Opana addiction requires residential treatment at least through the detoxification process. After that, the patient will be able to choose between remaining at the facility for further treatment or switching to outpatient care. Patients who are worried about relapsing should stay at the center until they are confident in their abilities.
- Long-Term or Short-Term: Because opiate addiction is difficult to break away from, long-term treatment is recommended. However, it depends on how severe the addiction is. Ultimately, the patient can have a preference but should defer to the guidance of their doctor.
- Treatment Goals: The user should have specific goals in mind which go beyond getting off the medication. Common goals for patients in drug rehabilitation centers are developing better self-control, learning to focus more on important relationships, and living a healthier life all around.
- Types of Therapy: No two rehabilitation centers are the same in their approach to treating drug addiction. Some centers take a highly clinical approach, while others create a real community within the treatment center. Some focus on athletics, while others focus on nature. Some use pharmaceuticals throughout the process, while others only use them during detox. The user needs to know what they want regarding the therapy offered.
Because users often want to stop abusing Oxymorphone, they can feel rushed to choose a center. Although getting off the medication as soon as possible is important, so is finding the right facility to be treated at. By speaking with addiction specialists, selecting the correct rehab for the Opana addiction facility should not take long.
Opana Rehabilitation Allows User to Live Healthier Lives
Addiction is a topic that makes many people uncomfortable, and that makes admitting there is a problem difficult to do. However, there is no shame in suffering from addiction, especially when seeking treatment. Opana addiction treatment centers do not judge the fact that a user is addicted or how that addiction came to be; they only seek to help.
No matter the path that led to addiction, there is a road to recovery. Opana addiction help should be sought before it is too late.
Oxymorphone is very addictive and difficult to stop. Not only is relapse likely when quitting without help, but the withdrawal process can be tough. Professional Opana addiction treatment ensures the user can quit the drug safely while developing the skills and perspective needed to remain in their future recovery.
Hope Without Commitment
Find the best treatment options. Call our free and confidential helpline
Most private insurances accepted
Find Drug Rehabilitation Centers Near You Anywhere In the US
Addiction Resource team has compiled an extensive list of the top drug rehabilitation facilities around the country. Use our locator tool to find the best centers near you.
Page Sources
- Kapila, A., Chhabra, L., Chaubey, V. K., & Summers, J. (2014). Opana ER abuse and thrombotic thrombocytopenic purpura (TTP)-like illness: a rising risk factor in illicit drug users. Case Reports, 2014, bcr2013203122.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3947999/
- Berner, T., Thomson, H., Hartry, A., Puenpatom, R. A., Ben-Joseph, R., & Szeinbach, S. L. (2011). A comparison of daily average consumption of Oxycodone controlled-release (OxyContin CR) and oxymorphone extended-release (Opana ER) in patients with low back pain. Pharmacy and Therapeutics, 36(3), 139.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3086105/
- Babalonis, S., Lofwall, M. R., Nuzzo, P. A., & Walsh, S. L. (2016). Pharmacodynamic effects of oral Oxymorphone: abuse liability, analgesic profile and direct physiologic effects in humans. Addiction Biology, 21(1), 146-158.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4383736/
- Boyle, K. L., & Rosenbaum, C. D. (2013). Oxymorphone insufflation associated with acute sensorineural hearing loss: case files of the University of Massachusetts medical toxicology fellowship. Journal of Medical Toxicology, 9(2), 179-183.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3657028/
- Mehmood, H., Khan, M., Marwat, A., Joshi, M., & Malhotra, V. (2018). Opana ER (Oxymorphone)–Induced Thrombotic Microangiopathy: An Atypical Presentation in a Patient With Hepatitis C. Journal of investigative medicine high impact case reports, 6, 2324709618756423.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5802609/
- Sloan, P. (2008). Review of oral Oxymorphone in the management of pain. Therapeutics and clinical risk management, 4(4), 777.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2621383/
- Rubino, M., Summers, K. H., Puenpatom, A., Fu, C., Ohsfeldt, R. L., & Ben-Joseph, R. H. (2011). A comparison of daily average consumption (DACON) of oxycodone and oxymorphone long-acting oral tablets. Journal of Managed Care Pharmacy, 17(5), 367-376.https://www.ncbi.nlm.nih.gov/pubmed/21657806
- Schoedel, K. A., McMorn, S., Bijan Chakraborty, M., Potts, S. L., Zerbe, K., & Sellers, E. M. (2011). Positive and negative subjective effects of extended-release Oxymorphone versus controlled-release Oxycodone in recreational opioid users. Journal of opioid management, 7(3), 179-192.https://www.ncbi.nlm.nih.gov/pubmed/21823549
- Schoedel, K. A., McMorn, S., Chakraborty, B., Zerbe, K., & Sellers, E. M. (2010). Reduced cognitive and psychomotor impairment with extended-release Oxymorphone versus controlled-release Oxycodone. Pain Physician, 13(6), 561-573.https://www.ncbi.nlm.nih.gov/pubmed/21102969
- National Institute on Drug Abuse, Principles of Drug Addiction Treatment: A Research-Based Guide (Third Edition) Types of Treatment Programs, https://www.drugabuse.gov/publications/principles-drug-addiction-treatment-research-based-guide-third-edition/drug-addiction-treatment-in-united-states/types-treatment-programs
- Adams, M. P., & Ahdieh, H. (2005). Single-and multiple-dose pharmacokinetics and dose-proportionality study of oxymorphone immediate-release tablets. Drugs in R & D, 6(2), 91-99.http://www.ncbi.nlm.nih.gov/pubmed/15777102
- U.S. National Library of Medicine, Medline Plus, Oxymorphone, https://medlineplus.gov/druginfo/meds/a610022.html
- U.S. Food & Drug Administration, FDA NEWS RELEASE, FDA requests removal of Opana ER for risks related to abuse, June 08, 2017, https://www.fda.gov/news-events/press-announcements/fda-requests-removal-opana-er-risks-related-abuse
- U.S. Food & Drug Administration, Oxymorphone (marketed as Opana ER) Information, https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/oxymorphone-marketed-opana-er-information
- Craig, D. S. (2010). Oxymorphone extended-release tablets (Opana ER) for the management of chronic pain: a practical review for pharmacists. Pharmacy and Therapeutics, 35(6), 324.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2888551/
- Azadfard M, Huecker MR, Leaming JM. (2020) Opioid Addiction. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK448203/
- Abuse, S. (2015). Key substance use and mental health indicators in the United States. National Survey on Drug Use & Health. SAMHSA.https://www.samhsa.gov/data/sites/default/files/NSDUH-FFR1-2016/NSDUH-FFR1-2016.htm#sud10


 Reviewed by:
Reviewed by:  Written by:
Written by: 
 FindTreatment.gov
FindTreatment.gov