Vilazodone is a powerful antidepressant belonging to the SSRI (selective serotonin reuptake inhibitor) group of drugs that helps people overcome major depressive disorder. While it is generally suitable for most depression patients, Viibryd side effects can present a significant obstacle to others. To benefit from treatment, patients should know what negative impacts the use of this medication can have and how to cope with them. It’s also crucial to learn when adverse effects may require medical intervention.
Table Of Contents:
In short-term clinical studies (those that do not exceed 4 months), it was found that antidepressants such as Viibryd increased the risk of suicidal thoughts in patients up to 24 years of age. However, it is unknown whether this risk applies to long-term use. What has been proven, though, is that drugs such as Viibyrd can be effective in preventing depression relapse in adults with major depressive disorder. Patients being treated with Vilazodone, especially those in their first few months of treatment, should be monitored closely for they can possibly develop suicidal thoughts, behavioral changes, and other serious adverse effects.
Viibryd side effects go awaywithin a few days or weeks after a person begins taking this medication. If they persist for longer, it’s necessary to contact a doctor.
What Are The Most Common Side Effects Of Vilazodone?
Among the 869 adult participants of two placebo-controlled studies, 7.1% or nearly 62 patients discontinued therapy due to various side effects of Viibryd. The most common of them being diarrhea, nausea, vomiting, and sleep problems. Based on clinical data from these trials, The National Center for Biotechnology Information (NCBI) reported in 2012 that 28% of patients who received Vilazodone treatment experienced diarrhea, while 23% experienced nausea, 6% experienced insomnia, and 5% experienced vomiting.
Diarrhea and vomiting can be particularly dangerous when they last for longer than two days. These side effects of Viibryd can cause severe dehydration in a person. If diarrhea persists, the patient should contact their doctor immediately. Negative symptoms that may require attention also include high blood pressure and brain zaps.
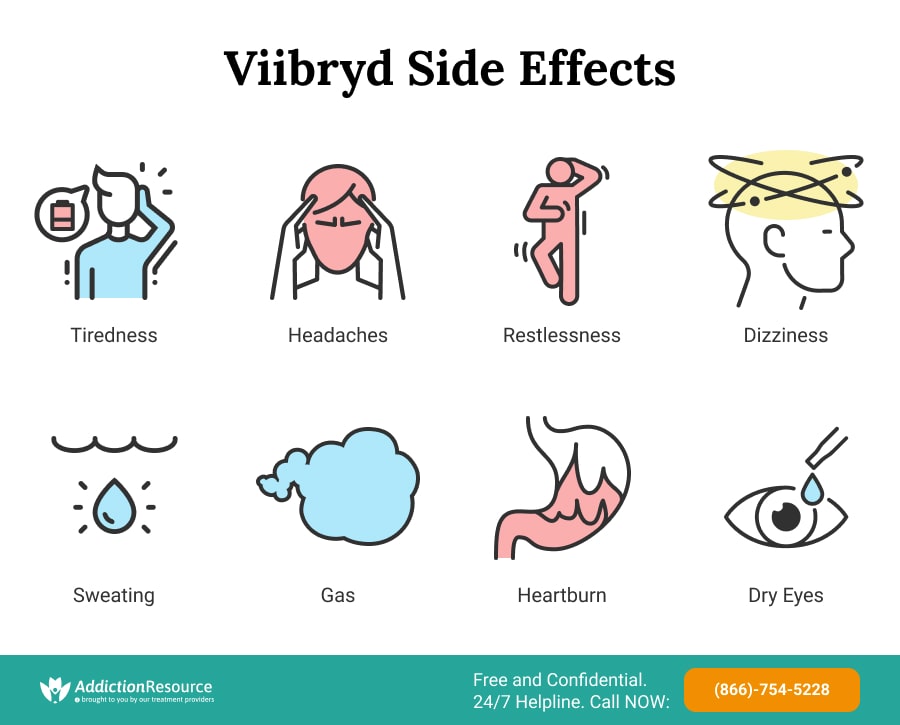
Other Common Viibryd Side Effects Include the Following:
- Tiredness
- Various sexual abnormalities
- Headache
- Restlessness
- Minor dizziness
- Sweating
- Gas
- Heartburn
- Dry mouth
- Changes in appetite
- Joint pain
- Unusual dreams
- Tinnitus
- Taste distortions
- Heartburn
- Dry eyes
To summarize, as an SSRI (selective serotonin reuptake inhibitor) antidepressant, Viibryd side effects are usually mild and go away quickly on their own. The most common adverse reactions to this medication include diarrhea, nausea, and Viibryd and sleep problems. If a person experiences diarrhea or vomiting for more than two days, it’s necessary to seek medical help, as both conditions can cause serious dehydration.
Rare Side Effects Of Vilazodone
Some adverse effects are incredibly rare, although it’s still possible for them to occur. They vary in severity, and some of them can be serious. Rare Viibryd side effects that do not go away quickly such as itching and rashes might indicate that the person is allergic to the medication and needs to find an alternative. Allergies can be serious and potentially life-threatening, and it’s important to watch out for signs of them. Other rare reactions of the medicine may include:
- Hair loss
- Unusual bleeding and bruising
- Pain or tingling in the hands or feet
- Difficulty breathing
- Tremors
- Seizures
- Agitation
- Hallucination
- Anxiety
- Panic attacks
- Mania
- Hyponatremia
- Coordination problems
- Serotonin syndrome
The FDA also warns against using the medication in people with pancreatic cancer and other health issues connected to the pancreas. In a dose as small as 20mg, symptoms can include:
- Acute pancreatitis
- Fever
- Upper abdominal pain
- Rapid pulse
If any of the above symptoms do not go away or are becoming a bother, seek a health professional’s advice. Patients on Vilazodone also sometimes experience hair loss and various sexual problems, including decreased libido and erectile dysfunction. Mental side effects of Vilazodone can include anxiety, mania, and nightmares. Contacting a person’s doctor and correcting the course of treatment can help reduce the negative impacts and improve the patient’s condition.
How Long Do Viibryd Side Effects Last?
Viibryd side effects go away within a few days or weeks after a person begins taking this medication. Most frequently, they cause mild discomfort and resolve without additional care when the patient adjusts to the drug. Such conditions as diarrhea, vomiting, and nausea go away after a couple of days. If they persist for longer, it’s necessary to contact a doctor.
Some psychological risks, such as depression and the lack of energy, may need several weeks to start improving. Vilazodone’s effects on one’s mental health may include severe depression when the drug is stopped abruptly. Sudden discontinuation of the drug also increases the risk of suicidal thoughts and behaviors. However, when a patient continues taking the medication as directed by their doctor, the adverse reactions usually diminish and eventually stop.
Vilazodone has a lower rate of negative sexual symptoms compared to other antidepressants, but still, Viibryd sexual side effects can be present in both men and women taking the drug.
Weight Changes On Vilazodone
Compared to other antidepressants, Viibryd is one that exhibits a lower incidence of effects on one’s weight. Information obtained from clinical studies suggest that both Viibryd and weight gain and Viibryd and weight loss do not have significant clinical relation. Weight changes will not be significant when patients use the drug as prescribed. However, if Vilazodone treatment is suddenly stopped or if a patient does not comply with their doctor’s instructions, reactions involving weight can be more noticeable. It’s also worth noting that Vilazodone’s interaction with other medications one is taking may cause changes in weight. Other factors such as dosage and duration of treatment may affect weight gain and weight loss as well.
Does Viibryd Cause Weight Gain?
One of the most feared long-term impacts of Vilazodone is weight gain. Depression frequently interferes with healthy eating patterns, forcing people to either overeat or disregard the need for nourishment. But as mentioned above, the relation between Viibryd and weight gain is not really of great significance. Based on clinical data, only about 1% and 2% of patients taking 20mg per day and 40mg per day of Vilazodone, respectively, had seen an increase in their weight. Weight gain may still be observed, though. For instance, noncompliant patients or those who suddenly stop taking the drug are more at risk of weight gain.
Does Viibryd Cause Weight Loss?
The relation between Viibryd and weight loss is also considered clinically insignificant – weight loss during Vilazodone treatment is very rare. A study in 2011 found that only 1.4% of patients experience 7 or more percent decrease in their body mass during treatment. Patients who struggle to stabilize their eating habits may find Vilazodone insufficient, as it is not designed to produce significant weight changes.

Sexual Side Effects Of Vilazodone
Vilazodone has a lower rate of negative sexual symptoms compared to other antidepressants, but still, Viibryd sexual side effects can be present in both men and women taking the drug. It’s not very common for Viibryd to negatively affect patients sexually, and even if it did, studies suggest that sexual-function-related adverse events caused by the drug were generally of low magnitude and not statistically significant. The rate of sexual adverse reactions associated with it is a little higher than placebo, though. Information derived from 4 placebo-controlled 2 to 2.5-month studies and an open-label 13-month study showed that common sexual adverse effects occurred in over 2% of Vilazodone patients. These common reactions include abnormal orgasm and anorgasmia, decrease in libido, and erectile dysfunction and ejaculation disorder in males.
Vilazodone Sexual Side Effects in Men
The 5 studies previously mentioned, found that men taking a higher dose of the drug were more at risk of Viibryd sexual side effects. Clinical information showed that erectile dysfunction didn’t occur in male participants who took 20mg of the drug per day, but about 3% of those who took 40mg per day were affected. Decrease in libido and ejaculation disorder also affected more men who took 40mg per day than men who took 20mg per day.
Decreased libido and ejaculation disorder occurred in 4% and 2% of the 40mg population, respectively, while only 3% and 1% of the 20mg population suffered from the same. Abnormal orgasm and anorgasmia, on the other hand, occurred in both populations equally at 2%.
Vilazodone Sexual Side Effects in Women
Viibryd sexual side effects occurred less in women than in men, and studies found no difference between how different dosages affected females. There were equal numbers of females who experienced sexual function effects in both 20mg-taking- and 40mg-taking-population. 1% of each population was affected by abnormal orgasm and anorgasmia, while 2% of each experienced a decrease in libido.
The sexual side effects of Viibryd tend to decrease over time, but if they persist and cause significant distress, the affected patient should contact their doctor or a health professional to discuss possible solutions.
The Influence Of Vilazodone On People’s Sleep Patterns
Viibryd and sleep can have a disruptive relationship in some patients. The most common side effects of Viibryd that manifest in sleep issues include sleep paralysis, nightmares, night sweats, and hypersomnia or insomnia – the most commonly reported Viibryd and sleep-related symptom. Normally, these adverse reactions are mild and resolve on their own after a couple of weeks.
Changes in sleep cycle are prevalent in people experiencing withdrawal symptoms – some of them experience excessive tiredness during the day and yet are having trouble going to sleep at night.

Viibryd During Pregnancy & Breastfeeding
As for the relation of Viibryd and pregnancy, there is currently no well-controlled human data regarding the matter. Due to the inadequacy of studies, the risks of major complications and miscarriage are unknown. There is also no human data regarding the presence of Viibryd in human milk, and so, its effects on milk production and the breastfed child are unknown. That said, it’s best to talk to a health professional for concerns about Viibryd and pregnancy as well as breastfeeding.
Can A Woman Take Viibryd While Pregnant?
The Viibryd and pregnancy category is C. It means that vilazodone has proved to be harmful to a degree in animal studies, but there isn’t enough data to gauge its hazardousness in humans. However, studies suggest that the use of Vilazodone during a pregnant woman’s third trimester may put their child at an increased risk for complications such as seizures, cyanosis, persistent pulmonary hypertension, respiratory distress, tremor, and feeding difficulty, among others. Again, one should talk to their doctor and carefully assess if the drug’s potential benefits outweigh the potential risks.
Can A Woman Breastfeed On Viibryd?
Since there’s no data that could answer whether or not Viibyrd is passed into human milk, it’s best to only breastfeed if the potential benefits outweigh the potential adverse effects. In studies involving rats, though, traces of Vilazodone were found in their milk. If one is breastfeeding or is planning to breastfeed, they should talk to a medical professional and consider the benefits of breastfeeding, their need for the drug, and the potential risks Viibryd may have on the breastfed child.
Minimizing Adverse Reactions of Viibryd
While most of the key adverse reactions that may stem from the use of Viibryd are not severe and usually go away after a couple of days or weeks, it’s still important to understand how the drug works. Gathering information about its possible adverse effects and the factors that may trigger them can help minimize the risk of them occurring.
Some Factors That May Influence Vilazodone’s Negative Effects Are:
- interactions with alcohol
- interactions with other drugs such as:
- Adderall
- CYP3A4 inducers
- CYP3A4 inhibitors
- Some drugs belonging to the monoamine oxidase inhibitors (MAOIs) class
- Some migraine medications
- Some antidepressants
- Some nonsteroidal anti-inflammatory drugs (NSAIDs)
- Interactions with herbs and supplements
The negative reactions may become worse when the medication is misused. Neglecting the prescription schedule may result in abuse and addiction. As soon as such behavior is noticed, it is critical to reaching professional rehab facilities. The staff in these centers will develop a personalized addiction treatment program to ensure fast and long-standing recovery.
Hope Without Commitment
Find the best treatment options. Call our free and confidential helpline
Most private insurances accepted
Page Sources
- American Society of Health-System Pharmacists. (2016, November 15). Vilazodone. From MedlinePlus: https://medlineplus.gov/druginfo/meds/a611020.html#side-effects
- Clayton, A., Kennedy, S., Edwards, J., Gallipoli, S., & Reed, C. (2013, October 10). The effect of vilazodone on sexual function during the treatment of major depressive disorder. The Journal of Sexual Medicine. From https://pubmed.ncbi.nlm.nih.gov/23216998/
- Cruz, M. (2012). Vilazodone HCl (Viibryd): A Serotonin Partial Agonist and Reuptake Inhibitor For the Treatment of Major Depressive Disorder. Pharmacy and Therapeutics, 28-31.
- Food and Drug Administration. (2017, January). HIGHLIGHTS OF PRESCRIBING INFORMATION: VIIBRYD (vilazodone hydrochloride) tablets. From Food and Drug Administration: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/022567s020lbl.pdf#page=17
- Hopkins, C. (2011). ACS chemical neuroscience molecule spotlight on viibryd (Vilazodone). ACS chemical neuroscience.
- Liebowitz, M., Croft, H., Kajdasz, D., Whalen, H., Gallipoli, S., Athanasiou, M., & Reed, C. (2011). The Safety and Tolerability Profile of Vilazodone, A Novel Antidepressant for the Treatment of Major Depressive Disorder. . Psychopharmacology bulletin.
- Mathis, M. (2016, September 6). Food and Drug Administration. From Food and Drug Administration: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2016/022567Orig1s019ltr.pdf

 Authored by
Authored by  Reviewed by
Reviewed by 

