Strattera is a commonly prescribed medication for children, teens, and adults. Generically known as atomoxetine, many people who use it do not have a good understanding of the drug’s health effects. Although it is not as addictive as other drugs, it still has the potential to be abused or cause adverse health effects. Besides, there are some atomoxetine interactions users should be aware of. In any case, people planning to take Strattera for ADHD need to know all information about this drug to know what to expect from the treatment.
Table Of Contents:
What Is Strattera?
Strattera is a prescription medication that has been sold in the United States since 2002. It is a selective norepinephrine reuptake inhibitor or SNRI, approved for use in patients aged six and older. It is the first non-stimulant medication approved for the treatment of ADHD. It is also not a controlled substance.

More About Antidepressants:
Atomoxetine may be prescribed to patients of different age groups. Since ADHD is a condition that affects both children and adults, using Strattera medication to manage the disorder among people of different ages is not uncommon. What may vary is the dose among patients. Although more research still needs to be undertaken to determine its efficacy in patients of certain age ranges.
How Does Strattera Work?
How this drug works refers to the process of inhibiting the reuptake of norepinephrine in the brain. It increases the levels of this neurotransmitter. It is achieved by selective inhibition of the presynaptic norepinephrine transporter. Reuptake is a normal mechanism by which the body controls how long a nerve signal lasts. However, if someone has significantly low levels of a neurotransmitter, reuptake can be a problem. The matter is that it restricts the volume of neurotransmitters being actively circulated in the brain.
What Is Strattera Used For?
The U.S. Food and Drug Administration has approved atomoxetine use for the treatment of ADHD in both children above six years and adult patients. In addition to these medical indications, the drug also has other off-label uses. It allows doctors to legally prescribe it for different age groups with a mixture of other conditions. A doctor may prescribe Strattera medication off-label for the treatment of mood and eating disorders, cognitive dysfunctions, and addiction to stimulant drugs.
Strattera For ADHD
Strattera ADHD effectiveness in patients may be attributed to the fact that the drug improves the overall ability of patients to pay attention, concentrate, and stay focused. As a non-stimulant drug, its use is generally considered less effective than psychostimulants in treating ADHD. However, it does cause potential Strattera side effects associated with psychostimulants as well.
Furthermore, a comparison of Strattera and Adderall, a stimulant drug, found that Adderall can be habit-forming. Hence it’s classified as a controlled substance, unlike atomoxetine. However, dependence on Strattera is also possible. One of the tradeoffs is that it may take longer for the drug to reach its full efficacy. That’s why patients often abuse it, trying to achieve the expected effects faster.

The initial improvements from Strattera ADHD were observed after one to four weeks after starting the treatment and clinically meaningful improvements after 10–26 weeks. But there is no study to indicate how long children and adults should be treated with this drug. It is generally agreed that treatment with Strattera for ADHD should be made over extended periods to achieve optimal efficiency. The medical doctor should periodically reevaluate the patient’s health and the prescribed dose.
Atomoxetine Interactions
One of the riskiest parts of taking any medication is the potential for interactions, which can cause unpleasant or dangerous symptoms. While Atomoxetine interactions are not more numerous than those found with other medications in its class, they are still dangerous, and users must be aware of them. Depending on the needs of the patient and their lifestyle, these risks could outweigh the atomoxetine benefits.
Atomoxetine And Alcohol
When mixing Strattera and alcohol, dangerous symptoms can result. Atomoxetine and alcohol come together to increase the effects of each substance, making it more likely that the user will become sick. Additionally, alcohol tends to weaken Strattera’s medical effectiveness.
Interactions with Marijuana
Atomoxetine is used in the treatment of marijuana dependence. It can also be combined with medical marijuana to treat ADHD. However, using it with weed recreationally is risky. Much like alcohol, cannabis can increase side effects while decreasing effectiveness. It also makes it more likely that the users will injure themselves.
Interactions with Zoloft
When a patient has both ADHD and depression or anxiety, they may be treated with atomoxetine hydrochloride and other antidepressants. However, combining Atomoxetine and Zoloft is not a good idea. When mixed with Zoloft, it can cause an irregular heart rhythm that can prove deadly in some patients.
Atomoxetine And Prozac
Atomoxetine interactions with Prozac are another risky combination. When it is mixed with Prozac, the blood concentration of atomoxetine increases. It causes severe side effects and dramatically increases the risk of overdose. It also produces an irregular heart rhythm that can be deadly.
Atomoxetine And Lexapro
As in the case with Zoloft, the combination with Lexapro can be deadly when combined due to their impact on the heart. The irregular heart rhythm produced is uncomfortable at best and fatal at worst. Users should avoid combining Lexapro with atomoxetine.

Of course, this is not a comprehensive list. Before taking the medication, users should consult with their doctor about all possible atomoxetine interactions and be open about any medications or other substances they are using. Always consult directly with a health specialist.
Can Strattera Be Abused?
The abuse of prescription medications is widespread. However, not all prescription medications lend themselves to abuse so easily.
When compared to other ADHD medications, the likelihood of Strattera medication abuse occurring is relatively low.
This is because it is not an amphetamine in the same way drugs like Adderall are. Instead, as an SNRI, it functions as an antidepressant.
With that said, antidepressants can still be abused. In general, doing this means taking a higher dose than prescribed or taking it more frequently than the user should. However, it can also be abused by mixing it with other medications and street drugs.
Is Strattera Addictive?
At this time, Strattera addiction is not considered to be possible. That is because studies have shown it is difficult to abuse and not result in physical addiction. However, this does not mean that people will never feel compelled to use the drug. People can develop a mental dependence on the drug if they think it brings them comfort. While the benefits of atomoxetine are critical to its use, they can also make patients fear life without it. As such, some might feel addicted even if they are not.
How Long Does Strattera Stay In One’s System?
Atomoxetine HCL half-life is roughly 5 hours. However, patients with a faster metabolism will see it eliminated sooner, and those with slower metabolisms will take longer to remove the drug from their system. As such, the Strattera half-life can vary between patients by several hours.
The half-life can also be affected by the performance of a patient’s organs. Approximately 80 percent of the dose will be removed by the kidneys and excreted in the urine. The rest is removed through the intestines. As such, any issues with the organs can result in slower removal and a longer half-life.
Does Strattera Show Up On Drug Tests?
A standard drug screening should not show the presence of atomoxetine hydrochloride in the system. However, that does not mean that it cannot produce a false positive. While this is rare, there is at least one confirmed case of atomoxetine triggering a false positive for amphetamines.
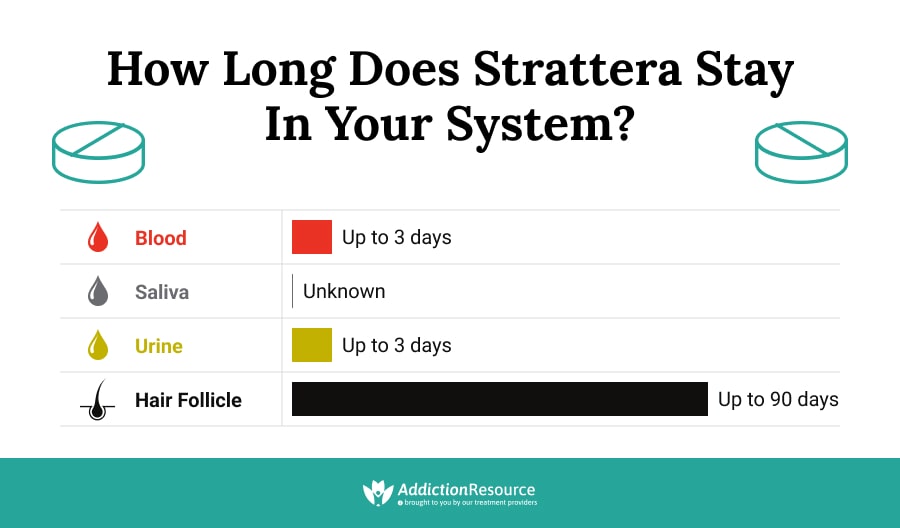
Generally speaking, only a Atomoxetine drug test should reveal the presence of the drug. However, it would be rare for a test to look for the presence of atomoxetine as it is not a common drug of abuse. With that said, users should still be aware of the detection times for the medication.
They Are as Follows:
- Urine: Up to 3 days
- Hair: Up to 90 days
- Blood: Up to 3 days
Users may need to disclose their use of Strattera medication before undergoing drug tests just if there is a false positive.
Stopping Strattera
Atomoxetine discontinuation should be done correctly. Unlike stimulant medications, this medication doesn’t affect dopamine levels. That is why in most cases stopping the drug will not cause acute Strattera withdrawal. A study on Strattera discontinuation stated that people with ADHD experienced condition symptoms worsening upon stopping the medication. However, no other adverse events were observed. It was concluded that tapering is unnecessary when stopping this drug.

Despite this, each case is unique; therefore, users should consult with medical professionals to take a safe and effective approach to end their use. Unlike stimulant ADHD medications, stopping atomoxetine generally does not produce a crash. However, it can still result in things like extreme changes in mood, suicidal thoughts, and extreme lethargy, which will be the symptoms of Strattera withdrawal.
Getting Help With Drug Misuse
Although Strattera ADHD medication is not on the list of controlled substances, it still poses a risk of dangerous interactions and abuse. Therefore, health professionals should educate patients, their families, and caregivers about the potential benefits of using the medication, as well as the associated risks. If someone has been misusing or abusing atomoxetine, they need to seek help in getting clean. Drug addiction rehab centers understand how to detox the patient and give them strategies to stay clean for the long term. The patient-centered addiction treatment programs are what most rehabs offer.
Frequently Asked Questions
Is Strattera a Stimulant?
Non-stimulant ADHD treatment options are becoming more prevalent, with atomoxetine being the most well-known in its class. Instead of stimulating the CNS, Atomoxetine, as a non-stimulant, increases norepinephrine in the brain. The way it works on the body is contradictory to the way stimulants act on the body.
Is Strattera a Controlled Substance?
It is not a controlled substance if looking at the DEA Schedule. Scheduled drugs are those in a class with a high potential for abuse. Atomoxetine is not considered a substance of abuse, and as such, it is not in any of the categories of controlled drugs.
How Long Does it Take for Strattera to Work?
Some people report small changes in hyperactivity and impulse control within two weeks of use. Generally, it may take 4 to 8 weeks for the drug to achieve maximum effectiveness.
Hope Without Commitment
Find the best treatment options. Call our free and confidential helpline
Most private insurances accepted
Page Sources
- Wietecha, L. A., Clemow, D. B., Buchanan, A. S., Young, J. L., Sarkis, E. H., & Findling, R. L. (2016). Atomoxetine increased effect over time in adults with Attention‐Deficit/Hyperactivity Disorder treated for up to 6 months: Pooled analysis of two double‐blind, placebo‐controlled, randomized trials. CNS neuroscience & therapeutics, 22(7), 546-557. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5069588/
- Wernicke, J. F., Adler, L., Spencer, T., West, S. A., Allen, A. J., Heiligenstein, J., ... & Michelson, D. (2004). Changes in symptoms and adverse events after discontinuation of atomoxetine in children and adults with attention deficit/hyperactivity disorder: a prospective, placebo-controlled assessment. Journal of clinical psychopharmacology, 24(1), 30-35. https://pubmed.ncbi.nlm.nih.gov/14709944/
- Budur, K., Mathews, M., Adetunji, B., Mathews, M., & Mahmud, J. (2005). Non-stimulant treatment for attention deficit hyperactivity disorder. Psychiatry (Edgmont), 2(7), 44. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3000197/
- FDA approves first generic Strattera for the treatment of ADHD. The US Food & Drug Administration. 2017. https://www.fda.gov/news-events/press-announcements/fda-approves-first-generic-strattera-treatment-adhd.
- Evans, E. A., & Sullivan, M. A. (2014). Abuse and misuse of antidepressants. Substance abuse and rehabilitation, 5, 107. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4140701/
- Upadhyaya, H. P., Desaiah, D., Schuh, K. J., Bymaster, F. P., Kallman, M. J., Clarke, D. O., ... & Allen, A. J. (2013). A review of the abuse potential assessment of atomoxetine: a nonstimulant medication for attention-deficit/hyperactivity disorder. Psychopharmacology, 226(2), 189-200. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3579642/
- STRATTERA (atomoxetine HCl): Package Insert. Eli Lilly and Company. 2002. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/21411_strattera_lbl.pdf
- Fenderson, J. L., Stratton, A. N., Domingo, J. S., Matthews, G. O., & Tan, C. D. (2013). Amphetamine positive urine toxicology screen secondary to atomoxetine. Case reports in psychiatry, 2013. https://www.ncbi.nlm.nih.gov/pubmed/23424703
- Tirado, C. F., Goldman, M., Lynch, K., Kampman, K. M., & Obrien, C. P. (2008). Atomoxetine for treatment of marijuana dependence: a report on the efficacy and high incidence of gastrointestinal adverse events in a pilot study. Drug and alcohol dependence, 94(1-3), 254-257. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3812929/





